|
University of Maryland School of Pharmacy, UNITED STATES Our computational epitope-mapping procedure is general and should thus prove useful for other viral envelope proteins whose structures have been characterized.Ĭitation: Sikora M, von Bülow S, Blanc FEC, Gecht M, Covino R, Hummer G (2021) Computational epitope map of SARS-CoV-2 spike protein. The protective glycan shield and the high flexibility of its hinges give the stalk overall low epitope scores. We find that the extensive and inherently flexible glycan coat shields a surface area larger than expected from static structures, highlighting the importance of structural dynamics. By mapping steric accessibility, structural rigidity, sequence conservation, and generic antibody binding signatures, we recover known epitopes on S and reveal promising epitope candidates for structure-based vaccine design. To identify possible antibody binding sites, we performed multi-microsecond molecular dynamics simulations of a 4.1 million atom system containing a patch of viral membrane with four full-length, fully glycosylated and palmitoylated S proteins. S is exposed on the viral surface and mediates viral entry into the host cell. So, Jianhui Huang, Niu Huang, and colleagues decided to look for other potential treatments that lack these flaws.The primary immunological target of COVID-19 vaccines is the SARS-CoV-2 spike (S) protein. 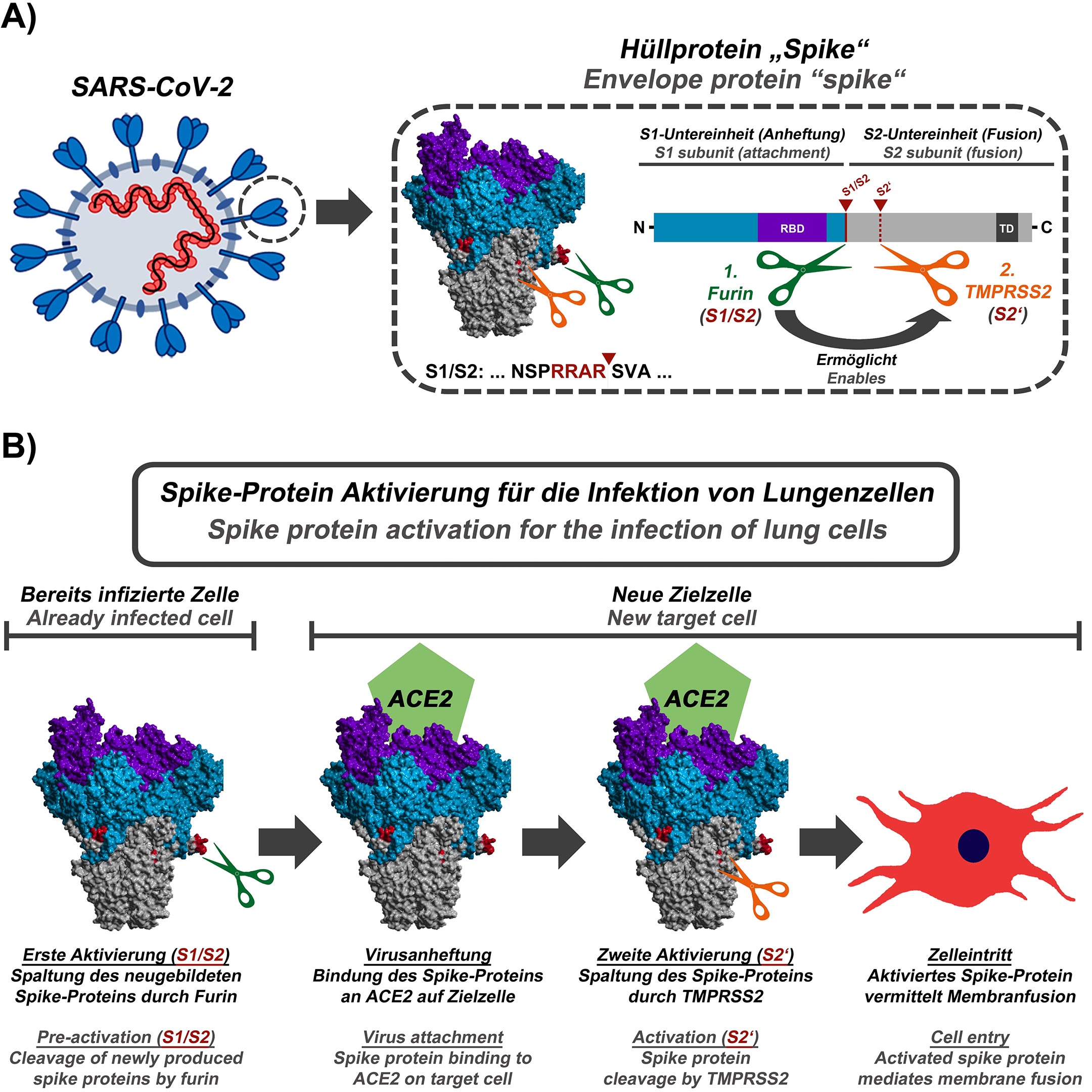
However, those compounds aren’t suitable treatments because they aren’t stable or they bind weakly. When this cranny is occupied by free fatty acids (FFAs) or a few other compounds, the protein remains locked in its closed, harmless configuration. One possibility is a pocket in the spike protein that has been dubbed the Achilles’ heel of the virus. To get around this problem, other, less mutation-prone parts of the spike protein could be targeted instead. That means vaccines and antibody therapies designed to target that fragment could become less effective as the virus mutates. However, some emerging variants of the coronavirus contain mutations in the RBD fragment. Antibodies contained in some COVID-19 therapies or stimulated by vaccines or infection target the RBD domain so it can’t bind to ACE2. In the “closed” structure, this RBD segment is tucked inside the spike protein and can’t bind to human cells. In their “open” structure, they expose a section known as the receptor-binding domain (RBD) so it can attach to the ACE2 protein on human cells. Spike proteins change shape when they attack a cell. Now, researchers report in ACS Central Science that they have discovered small molecules that successfully target other segments that mutate less. But those remedies gradually lose effectiveness when certain segments of the spike proteins mutate. 
Because of their important role in spreading infection, these spike proteins are one of the main targets for COVID-19 vaccines and treatments. The infamous spike proteins on the surface of SARS-CoV-2 help it bind to and enter human cells. Credit: Adapted from ACS Central Science 2023, DOI: 10.1021/acscentsci.2c01190 Using Small Molecules To Keep SARS-CoV-2 Closed for Business Some small molecules can lock SARS-CoV-2 spike proteins in a harmless closed configuration (left) instead of the infectious open configuration (right).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
